Bac2Bac
Over twenty million patients per year across the world develop respiratory failure and require mechanical ventilation in critical care units. A large proportion of these patients are at major risk of developing severe bacterial lung infections.
Often, chest x-ray changes demonstrate non-specific features that lead to the suspicion of a lung infection and trigger the administration of antibiotics.
We currently do not have bedside tests that can quickly and reliably diagnose infections at the bedside. Having these rapid tests available would be particularly helpful when a doctor needs to decide who should receive antibiotic treatments, which ones to use and when to stop them.
At present, many patients receive antibiotics because doctors do not have tests to guide their use and cessation with certainty. As a result, many patients are routinely prescribed antibiotics who probably do not need them or receive the wrong antibiotics for the type of infection they have. We need to find ways to safely decrease the use of antibiotics, thereby preventing the development of antibiotic resistance and ensuring infections are treated safely and effectively. We particularly need better ways to help doctors decide when to stop antibiotics.
This clinical study will find out how well a new technology works in intensive care patients requiring a breathing machine and who have a suspected or confirmed lung infection. We will pass a very fine microscope into regions of the lungs and deliver tiny amounts of chemicals which light up when they contact specific bacteria or inflammatory cells (see Figure 1).
We will also take small samples from those areas to determine the bacteria present. Knowing the type of bacteria tells us what antibiotics should be given. We will compare the results we get with methods currently used by doctors that take longer to provide results.
In the future, we hope that this technology can be adopted by health care organisations as a new cost effective method to diagnose and manage suspected lung infections in critically ill individuals, rationalising the use of antibiotics and improving outcomes for patients.
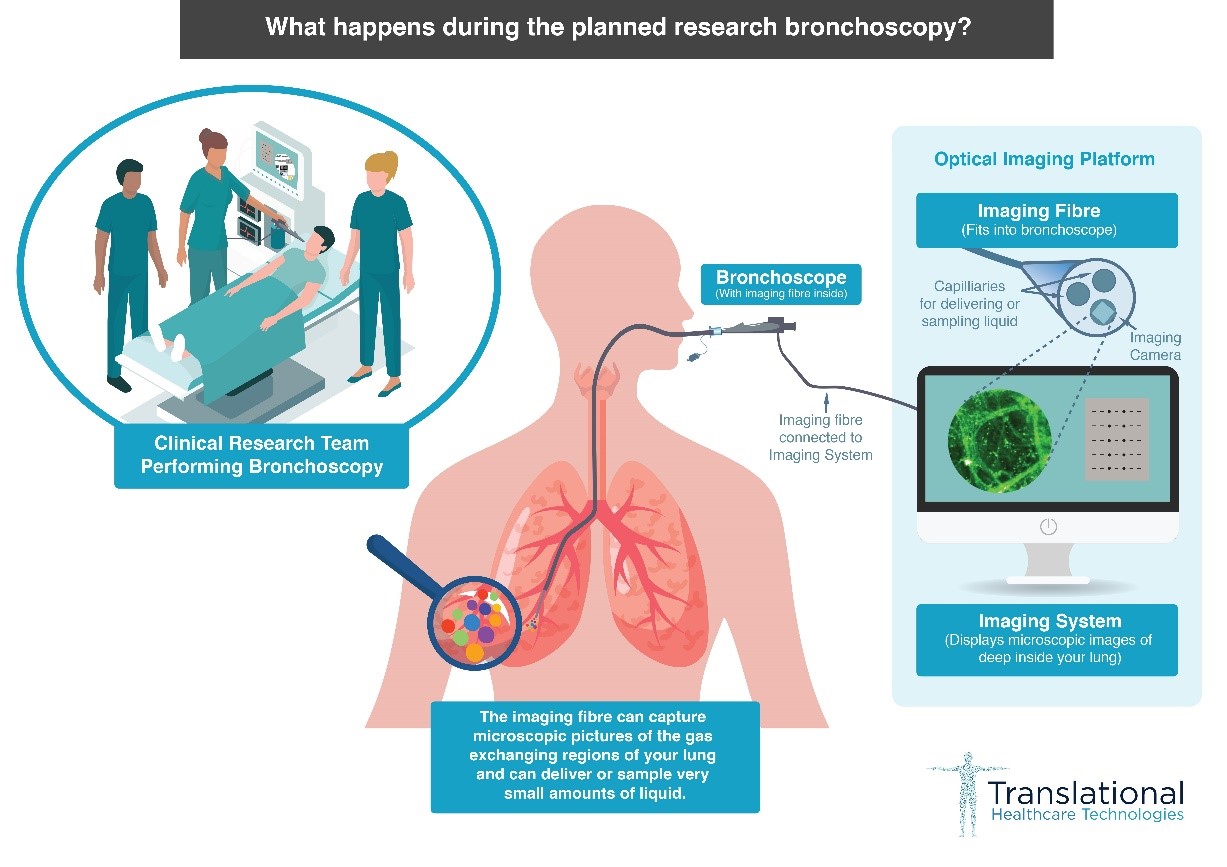
Figure 1: Diagram of what will happen during the BAC2BAC bronchoscopy.
This study will commence in Edinburgh in early 2023 and will test the safety and feasibility of the technology in approximately 12 patients. The study will then open recruitment in ICUs in Cambridge, London and Salford. The study will aim to recruit approximately 240 patients and will take approximately 2 years to complete.
